- April 4, 2025
- 1 second ago
Actics Medical - The digital revolution in treating a diabetic foot
The company has developed an AI-supported medical platform, with capabilities for continuous monitoring and risk warning with the potential to save millions of diabetes patients from amputations and death, every year.
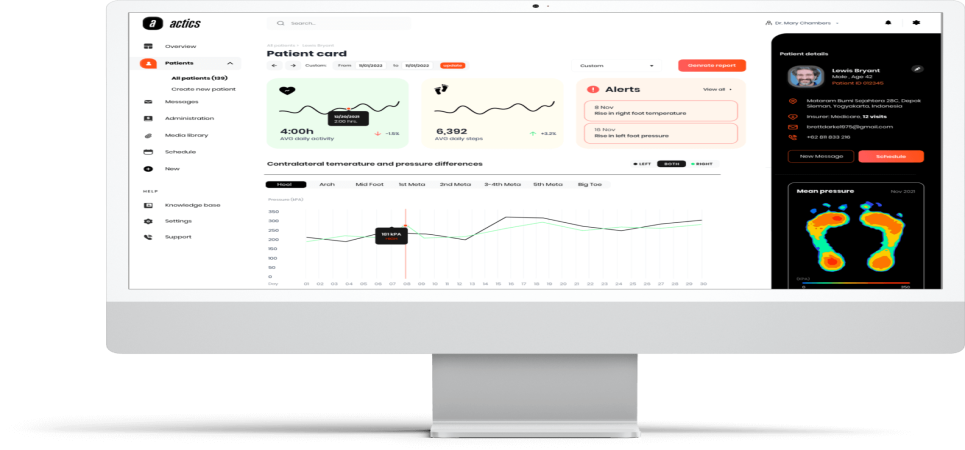
Actics Medical is the first company in the world to develop an advanced dynamic digital health platform for foot and body mobility problems, with smart insoles (Artificial Muscle) that combine bio-sensors and a custom-adjustable mechanism. The insoles enable continuous monitoring, accurate and advanced problem diagnosis, detection of various risks, and curative or preventive intervention.
The remote diagnostic and treatment capabilities of the company's products are based on deep learning (DL) analysis and artificial intelligence (AI) and enable to improve the lives of hundreds of millions of people who suffer from severe and chronic lower limb problems.
The company connects foot diagnosis and treatment to the digital healthcare system, a first-time capability, and enables the medical community to provide continuous, efficient, and effective service to a large number of patients. The digital health market enjoys accelerated growth estimated at a CAGR of 27.4% between the years 2023-2030.
The company's platform will provide innovative solutions for the numerous needs of large and diverse target audiences.
The company's first target audience includes the hundreds of millions of diabetes patients worldwide who are at acute risk of developing diabetic foot ulcers (DFU).
For this target audience, this is a life-saving solution.
Health Begins with the Feet
The feet play a vital role in our overall health. They are responsible for balance and mobility, autonomy, independence, and well-being. However, due to their complex anatomy, foot disabilities or deformities are growing more common and complicated. These problems impact the health of 20% to 40% of the population and impose a significant economic and functional burden on public health systems worldwide. Moreover, foot problems can be very complex to manage, and can quickly develop into acute, chronic, and irreversible conditions, particularly among diabetic foot patients at risk of developing DFU.
Actics Medical is developing broad-scope foot health solutions for a diverse target market that includes diabetes patients, neurological disorder patients (for example Parkinson's), sports injuries, body and skeletal mobility problems, balance, and more. The smart insoles have continuous monitoring and medical risk detection and diagnosis capabilities, as well as a built-in mechanism for accurate insole surface adjustment.
The combination of these features enables “correction and balancing” of pressure overloading in foot risk areas according to the patient's condition, to prevent the development of an ulcer or other problems, immediately and at any time.
This capability establishes the company as a pioneer in its field, with the ability to set the gold standard for diabetic foot treatment, and in the future in additional medical fields as well as in sports, mobility, and balance fields.
Why Invest in Actics Medical?
The Innovation and Technology
- The technology The company's technology combines real-time monitoring, cloud-based artificial intelligence, big data analysis, and artificial muscle to enable data collection, remote diagnosis, and dynamic adjustment of the insole surface for optimal and effective patient treatment.
- The company's products provide a solution for the vital health needs of hundreds of millions of people worldwide, and its technology has the potential for future use in sports and smart footwear products.
- The company conducted a POC of the technology and of the effectiveness of its solutions in two rigorous validation and effectiveness tests which showed excellent results.
Development and Achievements
- The company's first product, Actics OneTM, and the company plans to begin sales in Q4 2023.
- Actics Hybrid+TM insoles, the company's second product, are in advanced development stages, with clinical trials conducted at Hadassah Medical Center and NYU. Product commercialization is expected towards Q2 2024.
- The company was chosen as one of the TOP 20 companies at the Swiss DCB Open Innovation Challenge.
- The company participates in the “8400 Forum”, in which leading selected start-up companies in the health field receive professional accompaniment from senior managers in the industry.
- The company exhibited and delivered a lecture at OTWorld in Germany, the medical aid sector's largest trade show and world congress.
Intellectual Property and Grants
- The company received three grants from the Israel Innovation Authority (IIA).
- The company filed two patent registration applications that are in an advanced phase (national phase).
- The Hybrid product is categorized as FDA-registered Class 1, 510(k) exempt, the shortest and simplest registration track.
Target Markets
- The company is creating a “paradigm shift” in an outdated industry that is inefficient and seeks technological innovation, connecting the industry to the digital health and TeleHealth markets.
- The company's products belong to a growing group of products branded as Digital Therapeutics (DTx) and connected wearables and are part of the digital health market that is experiencing accelerated growth, with an estimated CAGR of 27.4% (2023-2030).
Team and Partners
- The company is headed by a diverse and experienced team in the medical, engineering, software, and business development fields.
- The company team is accompanied by physician opinion leaders, Prof. D. Armstrong from USC-California, and Prof. Lew Schon from NYU-New York, who also assist in presenting the company and its innovative products to the medical and business community.

Actics Technology
Antics technology is based on 2 patents and combines real-time monitoring, cloud-based artificial intelligence, and big data analysis, enabling data collection, remote diagnosis, and dynamic adjustment of the insole surface for optimal and effective patient treatment.

The company is developing and planning different levels of products to provide the broadest possible solutions to all the target audiences, addressing a wide range of needs and currently approved healthcare system budgets.
The company's first two products:
- One - a bio-mechanical insole with an adjustable built-in mechanism that enables high-level accurate customization to the patient's foot, in one visit to the orthopedist, the insole manufacturer, or the physiotherapist. The product will replace the current expensive and outdated treatment method that requires about 3 visits to a practitioner. Development was completed, was tested on tens of patients, and is ready for manufacturing and commercialization.
- Actics Hybrid+ - a revolutionary hybrid product that combines a digital-sensory system for continuous monitoring and irregularity alerts (pressure, temperature, motion) with a built-in adjustment mechanism. This enables immediate adjustment of the position on the insole to the updated foot condition and treatment of the clinical problem. Expected to reach the commercialization stage in Q2 2024.

Engineering validation tests of the measurement quality of the main risk indicators (pressure and temperature), which the company conducted at the Hadassah Medical Center Gait Laboratory, showed a 98% correlation compared to the measurement data of the accepted gold standard device. Furthermore, the company conducted clinical validation tests to monitor foot condition and deformity correction with tens of patients at Hadassah Medical Center and NYU (U.S.) and obtained excellent results.
In a letter summarizing the clinical validation trials, Prof. Lew Schon, a world-renowned foot and ankle surgeon and former president of the AOFAS, and Prof. David Armstrong, a surgeon, esteemed podiatry specialist, and healthcare policy advisor to governments and insurance companies, announced the results:

The company has begun to plan the “next generation” product that is based on Actics technology and combines new advanced technologies with an artificial muscle. This product will be an autonomous digital insole, combined with highly sensitive sensory capabilities and a smart polymer and nanorobotic-based adjustment mechanism.
This mechanism will simulate an artificial muscle and will enable electroactive deformation and construction of the insole surface structure within seconds to fully and accurately fit the patient's feet, with the ability to analyze and characterize the differences between the right and left foot.

Diabetes - Consumer Audience
One of the common complications of diabetes is damage to the nervous system (diabetic neuropathy), the muscle system, peripheral blood vessels, and loss of sensation in the lower limbs. There are over 200 million diabetes patients at risk of developing diabetic foot ulcers (DFU), of which about 15 million are in the U.S.
The annual national expenditure for DFU treatment in the U.S. alone is about $50 billion.
Diabetes patients who develop diabetic foot sores and ulcers (DFU) may develop severe infections that may even lead to gangrene and partial/full lower leg amputation. Studies prove that up to 80% of DFU cases can be prevented if early measures are taken to detect the development of the causes of the problem and to treat them quickly.
Actics technology provides a direct, accurate, and comprehensive solution for early detection of diabetic foot problems, enabling prevention in most cases as well as assistance in treatment.
General Orthopedics - Consumer Audience
A significant proportion of the world's population suffers from orthopedic problems that impact their everyday functioning, among them: flatfoot - characterized by a foot with little or no arch, or high arch foot - where the arch of the foot is abnormally high, as well as plantar fasciitis, metatarsalgia, severe corns, bunions, infections, Charcot and more. These conditions impair mobility and stability functions and the distribution of body weight on the feet, causing pain and differing degrees of mobility limitations. These physiological conditions can lead to additional problems such as knee and lower back problems.
Insurance companies do not include some of these conditions in their insurance policies and the expensive treatment costs are therefore incurred by the patient. Healthcare systems also provide only partial coverage and require co-payment. As a result, many avoid receiving medical treatment.
Insoles can provide a solution and alleviate limb fatigue or foot, thigh, knee, and even lower back pain. The company developed its products with the people suffering from these problems in mind. The aim is to simplify processes, reduce the number of treatments, and shorten the duration of each treatment, all at a cost that enables many patients to join those receiving treatment.
Practitioners in the Medical Community
The Actics Medical professional target audience includes podiatrists - foot specialists, orthopedists, insole manufacturers, and physiotherapists, who are in daily contact with patients suffering from foot and ankle problems and complications.
The specializing physicians are authorized to approve medical treatments and medical devices covered by the insuring entities: government health systems and private insurance companies.
The company presented its products in different development stages at professional conferences and in individual presentations to numerous practitioners in all treatment fields. The products garnered great interest among this target audience owing to two important aspects: the first, a significant medical-health solution for their patients, and second, adding a remote treatment system to their clinics, enabling them to provide a solution to a larger number of patients while increasing their revenues.
The Funding Entities
In most developed world countries both the government and private insurance systems approve and cover most of the medical treatment and product costs of diabetes patients. Insurers continuously examine new technologies and products that may contribute to improving the patient's condition, and especially to reduce their huge expenditures in this medical area. In the DFU field, the emphasis in recent years is on prevention. In the U.S., insurance companies have approved payment to courses of treatment (CPT codes) for remote monitoring, devices, and treatments that contribute to achieving this goal.

Activity Market
The company operates in the global digital health market that was valued at USD 223.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 27.4% from 2023 to 2030 and to reach USD 1,432.92 billion by 2030. Almost half a trillion dollars!
The significant growth driver in this market stems from the removal of numerous regulatory barriers in healthcare systems and changes in remote medicine accelerated by the coronavirus crisis, as well as from improved IP and VoIP communication infrastructures between physicians and patients. Furthermore, insurance companies adapted to these changes and provide insurance coverage for remote services.

Competition
The main competition facing Actics Medical is from the old and familiar methods and protocols according to which treatment only begins after an ulcer has developed and a severe foot health problem has arisen. The current medical treatment standard, which is based on clinic visits, has hardly achieved any success in efforts to prevent DFU.
In recent years, young companies (such as Orpyx, Podimetrics, and Siren) have developed digital products to monitor and warn of the developing danger of a pathology. These products enable partial monitoring and are not connected to a mechanism that enables preventive-integrative treatment and a rapid solution to acute risk conditions.
Moreover, their insoles are not customized and the patients must therefore order insoles that are manufactured and customized using outdated and cumbersome methods. This complexity results in the loss and waste of valuable medical and treatment time and increases the cost of the entire course of treatment.
Actics Medical offers a changed approach with a system that includes personally customized insoles for continuous monitoring that enables the detection of early warning signs, in addition to an integral mechanism for immediate treatment intervention - preventive and curative intervention in numerous risk cases.
Actics Medical Competitive Advantages
- Remote medicine: continuous and accurate remote diagnosis, monitoring, and treatment, in real-time, including detection and alert of physiological changes that may affect foot health.
- Prefabricated product: with high-precision customization for almost any problem requiring the use of a medical insole.
- Manufacturing costs: tens of percentages lower than those of the competitors.
- Built-in gait laboratory: for the first time the continuous monitoring in the insole captures real-time out-of-lab data during the user's everyday life as well, data which currently can only be obtained in gait laboratories. This offers significant cost-savings for physicians who treat patients and will not have to purchase and maintain expensive equipment and a large clinic.
- Integral dynamic adjustment mechanism: enables correction and personal customization of the insole surface (off-loading) to offload pressure and prevent the development of an ulcer, as well as pain or injury.
- Monitor effectiveness of preventive treatment: continuous update of patient preventive treatment compliance and persistence, and the clinical condition of the treated foot.
Business Model
The company has a B2B2C business model based on marketing partnerships with leading medical product distributors in the diabetes and orthopedic market that work with the target markets. These markets include diabetes physicians, orthopedic physicians, podiatrists, insole manufacturers, and physiotherapists.
In preparation and parallel, the company plans to establish its own representative office (in main concentrations of the target population - NY/east coast and California) to promote acquaintance with its products among the deciding entities - practitioners and insurance companies - and will make initial sales directly to the designated medical community.
There are two sales tracks in the diabetes and orthopedics market:
- The SaaS model is mainly customary in the diabetes market in which most patient costs are covered by insurance companies. In this track, most of the revenue to the company and the practitioners is received from ongoing monthly payments and the provision of private medical treatment. The basic monthly receipts - are according to four existing designated CPT codes for diabetes.
- The direct sale model that is usually applied to patients suffering from various orthopedic problems who are not fully covered or whose problem is not included under the coverage. These patients usually incur the cost of treatment, and our ability to significantly reduce this cost is therefore of great importance.



